Confocal Microscope 2
General Information
Technique
Key Instrumentation
Laser Scanning Confocal MicroscopeConfocal Microscope 2 is a Leica TCS SP8 unit mounted on a DMI8 inverted microscope. The instrument allows 3D imaging of fluorescently labeled systems such as biological membranes, cells, tissue, bacteria, hydrogels, colloidal systems. The Leica TCS SP8 microscope allows simultaneous acquisition of fluorescence emission from different dyes used to label different parts of the samples, using 8 laser lines for excitation and 5 independent detection channels. Among the five detection channels, three are highly sensitive GaAsp detectors particularly suitable for acquisition of low intensity emissions, for instance in case of samples affected by radiation damage or cytotoxicity. Fast image acquisition is possible thanks to the presence of a resonant scanner in addition to the conventional galvanometric scanner. In combination with a piezo actuated sample stage, this allows also acquisition of 3D volume scans down to the second timescale. This possibility allows to study samples in which relatively fast dynamic/kinetic processes are present. The microscope is also equipped with a Fluorescence Correlation Spectroscopy (FCS) module from PicoQuant that can be used to investigate fast (microsecond to millisecond) dynamical processes, like for example diffusion of molecules in bulk or in complex environments. The microscope is also equipped with a transmission detector for preliminary bright field imaging. An environmental chamber allows to control the temperature and relative humidity of the sample on the stage, and an additional fast heater directly applied to the microscope slide or Labtek well plates allows for temperature quenches.
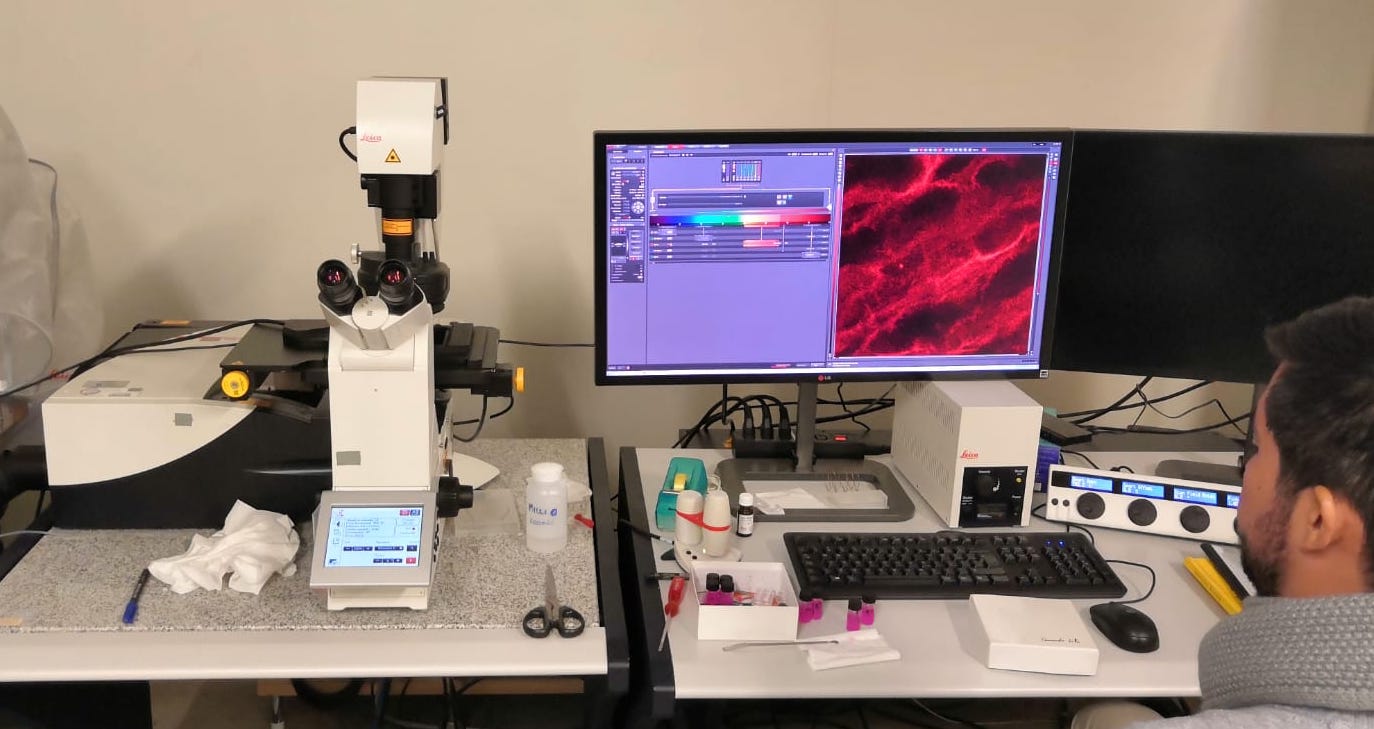
Technical description
Confocal Microscope 2 is a Leica TCS SP8 Laser Scanning Confocal Module mounted on a Leica DMI8 inverted microscope. The TSC SP8 module is equipped with a conventional galvanometric scanner, allowing low-noise acquisition of images up to 10fps (depending on image size), and a resonant scanner that allows fast acquisition of 512×512 pixel images at 31 frames per second (fps). Fluorescence excitation is obtained by illuminating the samples through 8 laser lines, while simultaneous fluorescence acquisition of 5 different dyes is possible thanks to 5 detectors, two of which are photo-multiplier tubes (PMT), one is a GaAsp hybrid detector and two are cooled GaAsp hybrid detectors, that are suitable for Single Molecule Detection. An additional PMT detector is present for transmission imaging. A motorized X-Y stage allows automatic acquisition of images at different sample locations and acquisition of large images with an automatic stitching procedure. The presence of a piezoelectric vertical (Z) focus scanner allows for fast 3D imaging, that combined with the use of the resonant scanner allows acquisition of 3D volume scans down to second timescales. The microscope is also equipped with a FCS PicoQuant module, that allows for spatially resolved FCS, FCCS, FRET and FRAP measurements. Additional available equipment: environmental chambers and tools for microfluidics.
Research areas and applications
The instrument allows performing time-resolved 3D imaging of fluorescently labeled samples of interest in a variety of fields, including live Cell Imaging, 3D Cell Culture, Membranes and Interfaces, Colloidal Systems, Polymers, Bacterial Motility, Gels, Medical Devices, Metals & Machine Engineering, Art conservation and restoration, Environmental Pollution. The large number of excitation lasers and detectors is suitable to investigate multiple-labeled samples. The presence of highly sensitive detectors allows investigation of samples labeled with dyes suffering photobleaching, and samples affected by radiation damage or cytotoxicity. The stage motorization is particularly useful for the investigation of large samples. The high speed of acquisition allows to study dynamic and kinetic properties of samples in 2D and 3D from tens of ms (2D) or s (3D) to long timescales, for example through particle tracking analysis of image or volume time series. The FCS and FCCS options are of particular interest for biological samples, to investigate diffusion processes on micro to millisecond timescales. Additional equipment for temperature scans and different relative humidity conditions is available.
Science highlights
R. Mastrangelo, D. Bandelli, L. Pensabene Buemi, P. Baglioni. Advanced Functional Materials. 34, 2404287 (2024). https://doi.org/10.1002/adfm.202404287
J. Cardellini, C. Dallari, I. De Santis, L. Riccio, C. Ceni, A. Morrone A, M. Calamai, F.S. Pavone, C. Credi, C. Montis, D. Berti. Nature Communications. 15, 7975 (2024). https://doi.org/10.1038/s41467-024-52205-9
A. Balestri, L. Gibot, H. Amenitisch, L. Cervelli, C. Montis, B. Lonetti, D. Berti. Colloids and Surfaces B: Biointerfaces. 231, 113532 (2023). https://doi.org/10.1016/j.colsurfb.2023.113532
G. Bassu, M. Izzi, A. Castellani, R.A. Picca, E. Fratini, M. Laurati, N. Cioffi. Materials Today Bio. 33, 101921 (2025). https://doi.org/10.1016/j.mtbio.2025.101921
R. Mastrangelo, G. Poggi, M. Laurati, E. Mascii, D. Bandelli, E. Fratini, P. Baglioni. Journal of Colloid and Interface Science. 694, 137745 (2025). https://doi.org/10.1016/j.jcis.2025.137745
Experimental team

- Marco Laurati
- CSGI-University of Florence
- Professor
