Gas Chromatography – Isotope Ratio Mass Spectrometry
General Information
Unit
CNR-ISPTechnique
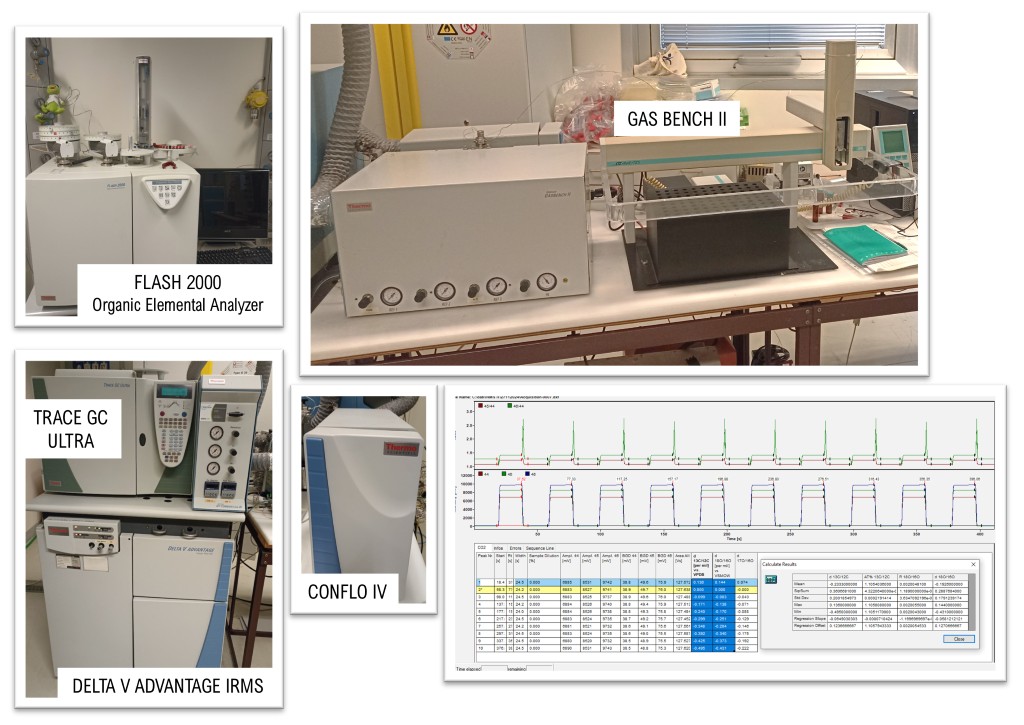
Key Instrumentation
Gas ChromatographyIsotope Ratio Mass Spectrometry (IRMS) is an analytical technique used for the determination of the relative quantities of stable isotopes of specific elements (C, H, O, N, S) following the quantitative combustion or pyrolysis of a solid or liquid sample, which is then analyzed in the gaseous phase.
The Thermo Fisher Scientific Delta V Advantage isotope ratio mass spectrometer is preceded by two different modules (Thermo Fisher Scientific Trace GC Ultra gas chromatograph and Flash 2000 Organic Elemental Analyzer) for sample processing, each equipped with both a combustion furnace and a pyrolytic furnace for the production of simple gases, which can then be separated and analyzed using the mass analyzer for the determination of the isotopic ratio of the elements C, H, O, N, S. The elemental analyzer allows for the determination of the percentage of C, H, N, and S present in unknown organic and inorganic compounds, both in the solid and liquid state, provided they are non-volatile. The GC allows for separation of single volatile compounds whose isotopic composition can be subsequently characterized through compound-specific isotope analysis (CSIA). The units that make up the hardware setup for GC-EA-IRMS are connected via the Thermo Scientific ConFlo IV Universal Interface, to manage different sample sources and gas flows in order to optimize the signal.
The Thermo Scientific GasBench II is a specialized, automated preparation device primarily used to analyze the stable isotope ratios δ of gaseous samples, most commonly: (i) CO2 released from water, carbonates, or dissolved inorganic carbon, (ii) water vapor and (iii) headspace gases.
Technical description
EA, GC-CSIA, and GasBench II Workflow
The Elemental Analyzer (EA) quantifies C, N, H, S, and O by combusting samples into gases (CO₂, N₂, H₂O, SO₂). These are refined via catalysts, separated by GC, and measured by a TCD to determine elemental percentages.
For Compound-Specific Isotope Analysis (CSIA), compounds separated by the TRACE 1310 GC are converted to simple gases in the GC-Interface III. This interface uses a backflush system to exclude solvents and protect the oxidation furnace, ensuring clean combustion without compromising GC performance.
The GasBench II handles headspace sampling for carbonates, water equilibration, and atmospheric gases. It utilizes helium as a carrier for GC separation and viscous flow transport, providing high-precision fractionation-free results. All systems connect through the ConFlo IV Interface, which automates gas flow adjustment and reference gas introduction to balance signal intensities.
IRMS Detection
Once purified gases enter the DELTA V IRMS, they undergo (i) ionization: electron bombardment creates positive ions; (ii) mass separation: the magnetic sector deflects ions based on mass; (iii) detection: multiple Faraday Cups simultaneously capture specific masses (e.g., 44, 45, and 46); (iv) analysis: 50V amplifiers measure the resulting currents. Software then calculates the final isotopic ratio (R = heavy/light).
Research areas and applications
Isotope Ratio Mass Spectrometry (IRMS), especially when coupled with preparation systems like Elemental Analyzers (EA), Gas Chromatography (GC), or a Gas Bench (GB), is a powerful tool with diverse applications across multiple research fields.
Main research applications include:
1) Environmental & Earth Sciences
• Paleoclimatology: analyzing δ¹⁸O and δD in ice cores or carbonates to reconstruct temperatures and hydrological cycles; studying δ¹³C in atmospheric CO₂ and tree rings to track the sources and sinks of greenhouse gases.
• Hydrology and Water Tracing: determining the origin and mixing of water bodies using δ¹⁸O and δD ratios.
• Biogeochemistry: using EA-IRMS (δ¹⁵N, δ¹³C) to study nutrient cycling in soils and sediments.
• Pollution Tracing: utilizing CSIA via GC-IRMS to identify the sources and degradation pathways of organic contaminants like pesticides.
2) Biology & Ecology
• Trophic Ecology: mapping food webs using δ¹⁵N and δ¹³C.
• Archaeology: reconstructing ancient diets and migration patterns by analyzing isotopes in bone collagen and tooth enamel.
3) Geology & Petrology
• Oil & Gas Exploration: performing GC-IRMS (CSIA) on individual hydrocarbons to correlate crude oil samples with their specific geological source rocks.
• Mineral Formation: measuring δ¹⁸O in minerals to determine temperature and environmental conditions during formation.
Science highlights
Battistel, S. Viva, C. Turetta, S. Cadamuro, E. Bonato, F. Giummolè, N. Lonoce, C. Barbante, S. Gelichi, Archaeol. Anthropol. Sci., 16 (8), 117 (2024).
https://doi.org/10.1007/s12520-024-02032-2Argiriadis, M. Bortolini, N.M. Kehrwald, M. Roman, C. Turetta, S. Hanif, E.O. Erhenhi, J.M. Ramirez Aliaga, D.B. McWethy, A.E. Myrbo, A. Pauchard, C. Barbante, D. Battistel, PLoS ONE, 16, e0254793 (2021). https://doi.org/10.1371/journal.pone.0254793
A. Kalashnikova, G. V. Simonova, O. V. Lyamkina, A. O. Pochufarov, J. Anal. Chem, 78,
1055-1061 (2023). https://doi.org/10.1134/S1061934823070079


